About Subnav
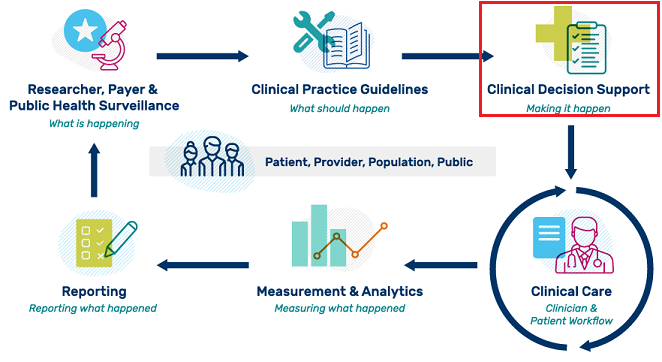
Clinical Decision Support (CDS) provides timely information to help inform decisions about a patient's care and has the ability to significantly impact improvements in quality, safety, efficiency, and effectiveness of health care. The Centers for Medicare & Medicaid Services (CMS) defines CDS as "health IT functionality that provides persons involved in care processes with general and person-specific information, intelligently filtered and organized, at appropriate times, to enhance health and health care.” CDS encompasses a variety of tools and processes to enhance decision-making in the clinical workflow. CDS is not simply an alert, notification, or explicit care suggestion. CDS helps clinical teams by supporting some routine tasks, warning of potential problems, or providing suggestions for the clinical team and patient to consider. CDS encompasses, but is not limited to
- Computerized alerts and reminders for providers and patients
- Condition-specific order sets
- Focused patient data reports, summaries, and dashboards
- Documentation templates
- Diagnostic support
- Contextually relevant reference information
CDS is not intended to replace clinician judgment, but rather to assist care team members in making timely, informed, and higher quality, evidence-based decisions, incorporating resources like clinical guidelines and best practices. According to Improving Outcomes with Clinical Decision Support: An Implementer's Guide, Second Edition, the CDS Five Rights model states improvements are achieved in desired healthcare outcomes if communicating
- The right information
- To the right people
- In the right CDS intervention formats
- Through the right channels
- At the right times in workflow
CDS and eCQMs are closely related, share many common requirements, data elements, and support health care quality improvement. The impact of a CDS intervention may be assessed with an eCQM.